DCDC Regulators
Triple-Output Step-Down Switching Regulator for Industrial and Portable Applications
A4490
Serving as a replacement part for the A4492 - Triple Output Step-Down Switching Regulator.
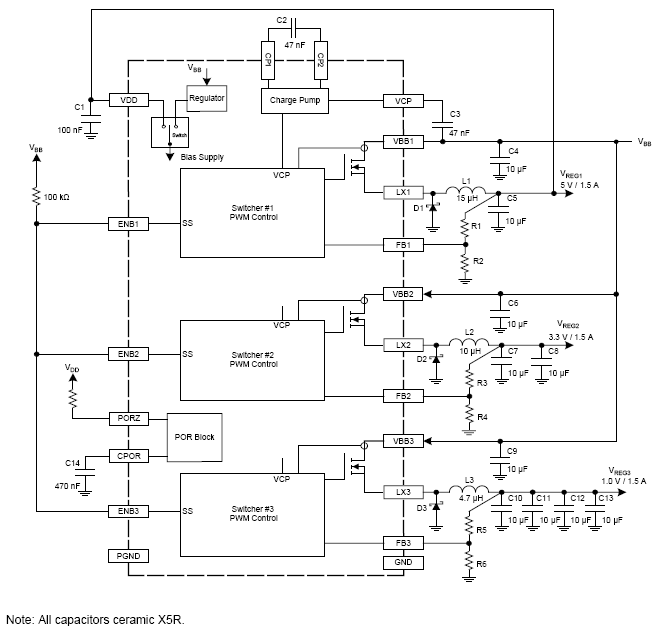
The A4490 efficiently powers multiple voltage rails with its three integrated buck converters, independent soft-start, and multiphase switching for reduced EMI. Ideal for industrial and portable equipment.
Product Details
Product Details
Top Features
- Three buck converters in a single IC
- Saves significant PCB space and component count vs single regulators
- 4.5 to 34 V input voltage range
- 550 kHz fixed frequency – stable with ceramic output capacitors
- Multiphase switching reduces EMI and input supply stress
- Independent control of each converter simplifies power sequencing
- Power-on-reset flag with adjustable delay
- Internal compensation
- 4 × 4 mm QFN Package, small PCB footprint